In general non-scientific terms, isotopes are slightly different types (or "versions") of atoms of the same element. (What is an Isotope ?) Meaning that isotopes are atoms of the same element with the same atomic number (same number of protons), but with a different atomic mass (distinct number of neutrons). How the isotopes of the same elements differ can also be seen through their chemical and physical properties. Typically the chemical properties of the isotopes are similar, whereas the physical properties are different due to their atomic structures.
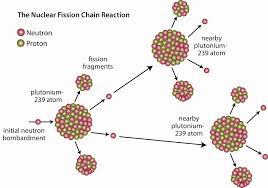
The isotopes relevant and most important to nuclear power are: 235U (Uranium-235) , 239Pu (Plutonium-239) and 233U (Uranium-233) (nuclearinfo.net) and all three are radioactive.
The isotopes relevant and most important to nuclear power are: 235U (Uranium-235) , 239Pu (Plutonium-239) and 233U (Uranium-233) (nuclearinfo.net) and all three are radioactive.